

收稿日期: 2019-04-01
网络出版日期: 2019-09-28
基金资助
国家自然科学青年基金资助项目(21601120);上海市自然科学基金资助项目(17ZR1410500)
Controlled synthesis of NiFe2O4/Co9S8 nanocomposites derived from metal-organic frameworks as efficient electrocatalysts for oxygen evolution reaction
Received date: 2019-04-01
Online published: 2019-09-28
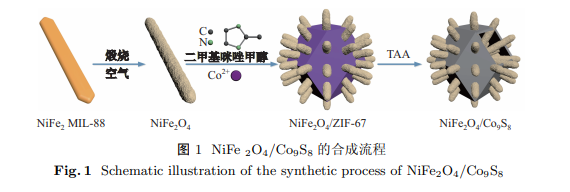
以金属有机骨架(metal-organic frameworks, MOFs)化合物为前驱体, 通过退火、硫化等后续处理方法成功制备了由 NiFe2O4 纳米棒与 Co9S8 空心球组装而成的杂化纳米结构. 该复合催化剂因具有较大的比表面积以及各组分间强力的协同作用, 在电催化析氧反应(oxygen evolution reaction, OER)中表现出了优异的催化活性. 在 1 mol/L KOH 电解质中, 当电流密度达到 10 mA/cm2 时, 仅需 290 mV 的过电位, 且 Tafel 斜率仅为 63.02 mV/dec. 此外, 1 000 次循环伏安(cyclic voltammetry, CV)测试后仍然具有良好的催化活性, 表明该复合催化剂具有优异的稳定性. 研究成果不仅为设计廉价、高效且稳定的析氧催化剂提供了设计思路, 也为其他新型纳米复合材料的合成和应用奠定了基础.
王尚岱, 黄守双, 胡张军, 陈志文 . 金属有机骨架衍生的 NiFe2O4/Co9S8复合催化剂的合成及其电催化析氧性能[J]. 上海大学学报(自然科学版), 2021 , 27(4) : 706 -716 . DOI: 10.12066/j.issn.1007-2861.2154
This paper reported the controlled synthesis of NiFe2O4/Co9S8 nanocomposites via annealing and sulfuration of metal-organic frameworks (MOFs). Due to the porous structure and the strong synergetic effects between the different composites, the as-prepared NiFe2O4/Co9S8 nanocomposite catalysts afforded an electrocatalytic oxygen evolution reaction (OER) with a current density of 10 mA/cm2 at a low overpotential of 290 mV and a small Tafel slope of approximately 63.02 mV/dec in 1 mol/L KOH. Moreover, the NiFe2O4/Co9S8 nanocomposite catalysts showed outstanding stability without obvious decline after 1 000 cycles of sequential cyclic voltammetry (CV) scanning. The findings not only provide a promising strategy for designing efficient and stable OER catalysts but also can be extended to synthesize other novel nanocomposite materials.

| [1] | Zhang Y, Zai J, He K, et al. Fe$_{3}$C nanoparticles encapsulated in highly crystalline porous graphite: salt-template synjournal and enhanced electrocatalytic oxygen evolution activity and stability[J]. Chemical Communications, 2018, 54:3158-3161. |
| [2] | Li X M, Qian T Y, Zai J T, et al. Co stabilized metallic 1T$_\mathrm d$ MoS$_{2}$ monolayers: bottom-up synjournal and enhanced capacitance with ultra-long cycling stability[J]. Materials Today Energy, 2018, 7:10-17. |
| [3] | Li X, Feng Z, Zai J, et al. Incorporation of Co into MoS$_{2}$/graphene nanocomposites: one effective way to enhance the cycling stability of Li/Na storage[J]. Journal of Power Sources, 2018, 373:103-109. |
| [4] | Li X, Zai J, Xiang S, et al. Regeneration of metal sulfides in the delithiation process: the key to cyclic stability[J]. Advanced Energy Materials, 2016, 6(19):1601056. |
| [5] | Yao L, Zhou L, Li S X, et al. Edge-rich MoS$_{2}$ nanosheets for high performance self-supporting Li-S batteries[J]. Energy Storage Science and Technology, 2019, 8(3):523-531. |
| [6] | Elizabeth I, Nair A K, Singh B P, et al. Multifunctional Ni-NiO-CNT composite as high performing free standing anode for Li ion batteries and advanced electro catalyst for oxygen evolution reaction[J]. Electrochimica Acta, 2017, 230:98-105. |
| [7] | Wu C, Kopold P, Van P A, et al. High performance graphene/Ni$_{2}$P hybrid anodes for lithium and sodium storage through 3D yolk-shell-like nanostructural design[J]. Advanced Materials, 2017, 29(3):1604015. |
| [8] | Liu X, Liu Y, Fan L Z. MOF-derived CoSe$_{2}$ microspheres with hollow interiors as high-performance electrocatalysts for the enhanced oxygen evolution reaction[J]. Journal of Materials Chemistry A, 2017, 5:15310-15314. |
| [9] | Ping L, Rodriguez J A. Catalysts for hydrogen evolution from the [NiFe] hydrogenase to the Ni$_{2}$P(001) surface: the importance of ensemble effect[J]. Journal of the American Chemical Society, 2005, 127:14871-14878. |
| [10] | Zeng K, Zhang D. Corrigendum to “recent progress in alkaline water electrolysis for hydrogen production and applications”[J]. Progress in Energy and Combustion Science, 2011, 37:631. |
| [11] | Walter M G, Warren E L, McKone J R, et al. Solar water splitting cells[J]. Chemical Reviews, 2010, 110:6446-6473. |
| [12] | Yu C, Huang H, Zhou S, et al. An electrocatalyst with anti-oxidized capability for overall water splitting[J]. Nano Research, 2018, 11:3411-3418. |
| [13] | Zhuang H, Xie Y, Tan H, et al. CoFe$_{x}$-CoFe$_{2}$O$_{4}$/N-doped carbon nanocomposite derived from in situ pyrolysis of a single source precursor as a superior bifunctional electrocatalyst for water splitting[J]. Electrochimica Acta, 2018, 262:18-26. |
| [14] | You B, Sun Y J. Hierarchically porous nickel sulfide multifunctional superstructures[J]. Advanced Energy Materials, 2016, 6(7):1502333. |
| [15] | Cui X, Xie Z, Wang Y. Novel CoS$_{2}$ embedded carbon nanocages by direct sulfurizing metal-organic frameworks for dye-sensitized solar cells[J]. Nanoscale, 2016, 8:11984-11992. |
| [16] | Liu Q, Zhang J. A general and controllable synjournal of Co$_{m}$S$_{n}$ (Co$_{9}$S$_{8}$, Co$_{3}$S$_{4}$, and Co$_{1-x}$S) hierarchical microspheres with homogeneous phases[J]. CrystEngComm, 2013, 15:5087. |
| [17] | Yu L, Yang J F, Lou X W. Formation of CoS$_{2}$ nanobubble hollow prisms for highly reversible lithium storage[J]. Angew Chem Int Ed Engl, 2016, 55:13422-13426. |
| [18] | Zhu Y, Song L, Song N, et al. Bifunctional and efficient CoS$_{2}$-C@MoS$_{2}$ core-shell nanofiber electrocatalyst for water splitting[J]. ACS Sustainable Chemistry & Engineering, 2019, 7:2899-2905. |
| [19] | Yang J, Zhu G, Liu Y, et al. Fe$_{3}$O$_{4}$-decorated Co$_{9}$S$_{8 }$ nanoparticles in situ grown on reduced graphene oxide: a new and efficient electrocatalyst for oxygen evolution reaction[J]. Advanced Functional Materials, 2016, 26:4712-4721. |
| [20] | Hao J, Yang W, Peng Z, et al. A nitrogen doping method for CoS$_{2}$ electrocatalysts with enhanced water oxidation performance[J]. ACS Catalysis, 2017, 7:4214-4220. |
| [21] | Song F, Hu X. Ultrathin cobalt-manganese layered double hydroxide is an efficient oxygen evolution catalyst[J]. Journal of American Chemical Society, 2014, 136:16481-16484. |
| [22] | Yeo B S, Bell A T. Enhanced activity of gold-supported cobalt oxide for the electrochemical evolution of oxygen[J]. Journal of American Chemical Society, 2011, 133:5587-5593. |
| [23] | Zhu H, Zhang S, Huang Y X, et al. Monodisperse M$_{x}$Fe$_{3-x}$O$_{4}$ (M=Fe, Cu, Co, Mn) nanoparticles and their electrocatalysis for oxygen reduction reaction[J]. Nano Letters, 2013, 13:2947-2951. |
| [24] | Li M, Xiong Y, Liu X, et al. Facile synjournal of electrospun MFe$_{2}$O$_{4}$ (M=Co, Ni, Cu, Mn) spinel nanofibers with excellent electrocatalytic properties for oxygen evolution and hydrogen peroxide reduction[J]. Nanoscale, 2015, 7:8920-8930. |
| [25] | Liu G, Wang K, Gao X, et al. Fabrication of mesoporous NiFe$_{2}$O$_{4}$ nanorods as efficient oxygen evolution catalyst for water splitting[J]. Electrochimica Acta, 2016, 211:871-878. |
| [26] | Yang H, Liu Y, Luo S, et al. Lateral-size-mediated efficient oxygen evolution reaction: insights into the atomically thin quantum dot structure of NiFe$_{2}$O$_{4}$[J]. ACS Catalysis, 2017, 7:5557-5567. |
| [27] | Liu J, Zhu D, Ling T, et al. S-NiFe$_{2}$O$_{4}$ ultra-small nanoparticle built nanosheets for efficient water splitting in alkaline and neutral pH[J]. Nano Energy, 2017, 40:264-273. |
| [28] | Indra A, Menezes P W, Sahraie N R, et al. Unification of catalytic water oxidation and oxygen reduction reactions: amorphous beat crystalline cobalt iron oxides[J]. Journal of the American Chemical Society, 2014, 136:17530-17536. |
| [29] | Landon J, Demeter E, Inoglu N, et al. Spectroscopic characterization of mixed Fe-Ni oxide electrocatalysts for the oxygen evolution reaction in alkaline electrolytes[J]. ACS Catalysis, 2012, 2:1793-1801. |
| [30] | Chen H, Yan J, Wu H, et al. One-pot fabrication of NiFe$_{2}$O$_{4}$ nanoparticles on $\alpha$-Ni(OH)$_{2}$ nanosheet for enhanced water oxidation[J]. Journal of Power Sources, 2016, 324:499-508. |
| [31] | Wu Z, Zou Z, Huang J, et al. NiFe$_{2}$O$_{4}$ nanoparticles/NiFe layered double-hydroxide nanosheet heterostructure array for efficient overall water splitting at large current densities[J]. ACS Applied Materials $\&$ Interfaces, 2018(10):26283-26292. |
| [32] | Huang G, Zhang F, Zhang L, et al. Hierarchical NiFe$_2$O$_4$/Fe$_2$O$_3$ nanotubes derived from metal organic frameworks for superior lithium ion battery anodes[J]. Journal of Materials Chemistry A, 2014, 2:8048-8053. |
| [33] | Wei J, Hu Y, Liang Y, et al. Graphene oxide/core-shell structured metal-organic framework nano-sandwiches and their derived cobalt/N-doped carbon nanosheets for oxygen reduction reactions[J]. Journal of Materials Chemistry A, 2017, 5:10182-10189. |
| [34] | Lin K Y A, Chang H A. Zeolitic imidazole framework-67 (ZIF-67) as a heterogeneous catalyst to activate peroxymonosulfate for degradation of Rhodamine B in water[J]. Journal of the Taiwan Institute of Chemical Engineers, 2015, 53:40-45. |
| [35] | Lin K Y, Chang H A. Ultra-high adsorption capacity of zeolitic imidazole framework-67 (ZIF-67) for removal of malachite green from water[J]. Chemosphere, 2015, 139:624-31. |
| [36] | Huang S, Meng Y, He S, et al. N-, O-, and S-tridoped carbon-encapsulated Co$_{9}$S$_{8}$ nanomaterials: efficient bifunctional electrocatalysts for overall water splitting[J]. Advanced Functional Materials, 2017, 27(17):1606585. |
| [37] | Bao J, Zhang X, Fan B, et al. Ultrathin spinel-structured nanosheets rich in oxygen deficiencies for enhanced electrocatalytic water oxidation[J]. Angew Chem Int Ed Engl, 2015, 54:7399-7404. |
| [38] | Cao X, Zheng X, Tian J, et al. Cobalt sulfide embedded in porous nitrogen-doped carbon as a bifunctional electrocatalyst for oxygen reduction and evolution reactions[J]. Electrochimica Acta, 2016, 191:776-783. |
| [39] | Mahala C, Sharma M D, Basu M. 2D nanostructures of CoFe$_{2}$O$_{4}$ and NiFe$_{2}$O$_{4}$: efficient oxygen evolution catalyst[J]. Electrochimica Acta, 2018, 273:462-473. |
/
| 〈 |
|
〉 |