

Structural and electronic properties of nitrogen and boron substitutions of C$_{64}$-graphyne: the first-principle calculations
Received date: 2018-09-21
Online published: 2018-12-23
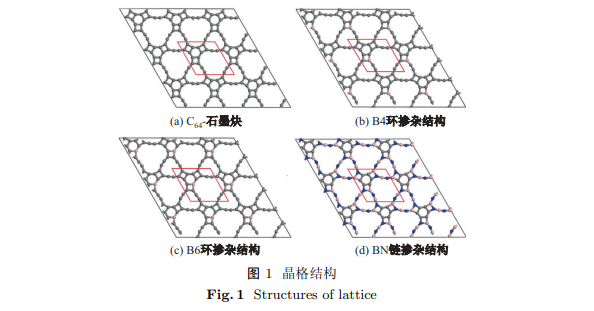
采用基于密度泛函理论的第一性原理计算方法, 对新型碳单原子层平面结构—C64-石墨炔的硼、氮掺杂结构进行研究. 当硼原子分别替代C64-石墨炔中碳六元环和四元环上的一个碳原子时, 获得稳定的四元环位B掺杂C64-石墨炔结构和六元环位B掺杂C64-石墨炔结构(简称B4环掺杂结构和B6环掺杂结构). 两种结构仍然表现为二维平面结构, 晶格常数分别为9.378×10-10和9.383×10-10 m; 当硼和氮原子交替替代C64-石墨炔中四元环和碳链上的碳原子时, 得到链位B, N掺杂C64-石墨炔稳定平面结构(简称BN链掺杂结构), 晶格常数为9.393×10-10 m. 单个硼原子掺杂的 B4 环掺杂结构和 B6 环掺杂结构均因掺杂使体系由半导体转变为金属. 硼、氮原子交替取代的BN链掺杂结构是带隙为2.56 eV的半导体.
李晖, 辛子华 . 硼氮掺杂C64-石墨炔材料几何及电子结构第一性原理[J]. 上海大学学报(自然科学版), 2020 , 26(5) : 816 -823 . DOI: 10.12066/j.issn.1007-2861.2091
By using the first-principles calculations based on density functional theory, the nitrogen and boron substitutions of a novel monolayer planar structure named C64-graphyne are studied. By substituting one nitrogen atom for a carbon atom in the hexatomic ring or tetratomic ring, two planar structures are obtained, which are named B4ringC-C64and B6ringC-C64 with the lattice parameter of 9.378×10-10 m and 9.383×10-10 m, respectively. Further investigation shows that a new structure named (BN)chain-C64 can be obtained by alternating substitution of boron and nitrogen atoms for carbon atoms in tetratomic ring and carbon chain. The doping with single boron atom induces the metal properties of two monolayer structures. The alternating substitution of boron and nitrogen atoms makes it act as semiconductor with band gap of 2.56 eV.

| [1] | Novoselov K S, Geim A K, Morozov S V, et al. Electric field effect in atomically thin carbon films[J]. Science, 2004,306(5696):666-669. |
| [2] | Neto A H C, Guinea F, Peres N M R, et al. The electronic properties of graphene[J]. Reviews of Modern Physics, 2009,81(1):109-162. |
| [3] | Nair R R, Blake P, Grigorenko A N, et al. Fine structure constant defines visual transparency of graphene[J]. Science, 2008,320(5881):1308. |
| [4] | Kuloglu A F, Sarikavak-Lisesivdin B, Lisesivdin S B, et al. First-principles calculations of Pd-terminated symmetrical armchair graphene nanoribbons[J]. Computational Materials Science, 2013,68:18-22. |
| [5] | Wu M, Cao C, Jiang J Z. Light non-metallic atom (B, N, O and F)-doped graphene: a first-principles study[J]. Nanotechnology, 2010,21(50):505202. |
| [6] | Wei D, Liu Y, Wang Y, et al. Synjournal of N-doped graphene by chemical vapor deposition and its electrical properties[J]. Nano Letters, 2009,9(5):1752-1758. |
| [7] | Sheng Z H, Gao H L, Bao W J, et al. Synjournal of boron doped graphene for oxygen reduction reaction in fuel cells[J]. Journal of Materials Chemistry, 2011,22(2):390-395. |
| [8] | Panchakarla L S, Subrahmanyam K S, Saha S K, et al. Synjournal, structure, and properties of boron and nitrogen doped graphene[J]. Advanced Materials, 2009,21(46):4726-4730. |
| [9] | Baughman R H, Eckhardt H, Kertesz M. Structure-property predictions for new planar forms of carbon: layered phases containing sp 2 and sp atoms[J]. The Journal of Chemical Physics, 1987,87(11):6687-6699. |
| [10] | Li G, Li Y, Liu H, et al. Architecture of graphdiyne nanoscale films[J]. Chemical Communications, 2010,46(19):3256-3258. |
| [11] | Zhang Y Y, Pei Q X, Wang C M. Mechanical properties of graphynes under tension: a molecular dynamics study[J]. Applied Physics Letters, 2012,101(8):081909. |
| [12] | Long M, Tang L, Wang D, et al. Electronic structure and carrier mobility in graphdiyne sheet and nanoribbons: theoretical predictions[J]. Acs Nano, 2011,5(4):2593-2600. |
| [13] | Ma S Y, Zhang M, Sun L Z, et al. High-temperature behavior of monolayer graphyne and graphdiyne[J]. Carbon, 2016,99(11):547-555. |
| [14] | Zhang J J, Xin Z H, Zhang J H, et al. Molecular dynamics study on the stability and properties of alpha-CGeyne[J]. Acta Physica Sinica, 2014,63(20):207303. |
| [15] | Liu J, Xin Z, Yan X, et al. Structural, phononic and electronic properties of Ge-doped $\gamma$-graphynes: a first-principles study[J]. Solid State Communications, 2017,258:38-44. |
| [16] | Yan X, Xin Z H, Zhang J J, et al. Molecular dynamics study on the structure and properties of silicon-graphdiyne[J]. Acta Physica Sinica, 2013,62(23):238101. |
| [17] | Singh N B, Bhattacharya B, Sarkar U. A first principle study of pristine and BN-doped graphyne family[J]. Structural Chemistry, 2014,25(6):1695-1710. |
| [18] | Kang B, Shi H, Wang F F, et al. Importance of doping site of B, N, and O in tuning electronic structure of graphynes[J]. Carbon, 2016,105:156-162. |
| [19] | Yun J, Zhang Z, Yan J, et al. First-principles study of B or Al-doping effect on the structural, electronic structure and magnetic properties of $\gamma$-graphyne[J]. Computational Materials Science, 2015,108(12):147-152. |
| [20] | Kim H, Kim Y, Kim J, et al. Computational searching for new stable graphyne structures and their electronic properties[J]. Carbon, 2016,98:404-410. |
| [21] | Song Q, Wang B, Deng K, et al. Graphenylene, a unique two-dimensional carbon network with nondelocalized cyclohexatriene units[J]. Journal of Materials Chemistry C, 2013,1(1):38-41. |
| [22] | Lu H G, Li S D. Two-dimensional carbon allotropes from graphene to graphyne[J]. Journal of Materials Chemistry C, 2013,1(23):3677-3680. |
/
| 〈 |
|
〉 |