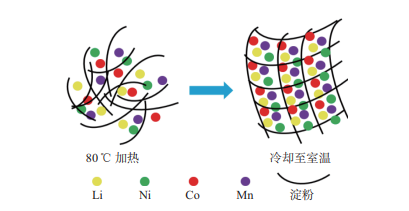
To improve the energy density of lithium-ion batteries, a high-nickel ternary cathode material LiNi0.8Co0.1Mn0.1O2 (NCM811) has attracted extensive attention. The raw materials for preparing NCM811 are uniformly dispersed in a high-temperature starch solution by utilizing the characteristic that starch forms a solution at high temperatures and condenses into a gel at lower temperatures. A gel with homogeneous decentralized metal ions was observed, and the NCM811 was prepared by calcining in an air atmosphere, as opposed to the conventional method that uses an oxygen atmosphere. The effects of starch content on the crystal structure, granule size, and electrochemical performance of electrode materials were examined. For 10% starch concentration, particle size was approx-imately 500 nm, coulombic efficiency was 82.88%, discharge specific capacity at 1.0 C was 146.0 mA·h·g−1, and capacity retention for 100 cycles at 1.0 C was 80.14%. The reduc-tion in particle size of the material was attributed to the space confinement effect of the starch gel and the inhibition of particle aggregation and growth during calcination. The small particle size accelerates the migration between electrode and electrolyte, reducing the transport distance of lithium ions. Because of the existence of amylum, the surface of the material contains residual carbon, which enhances the conductivity of the material, thus improving the electrochemical performance.
HU Yao, GONG Jianyang, YOU Wanli, LIU Hongjiang, CHEN Guorong, SHI Liyi,
. Preparation and electrochemical performances of LiNi0.8Co0.1Mn0.1O2 using an in situ gel system[J]. Journal of Shanghai University, 2023
, 29(2)
: 302
.
DOI: 10.12066/j.issn.1007-2861.2305


