

Journal of Shanghai University >
Preparation, phosphorus removal performance and mechanism of lanthanum oxide/expanded graphite composite adsorbents
Received date: 2018-05-17
Online published: 2021-07-22
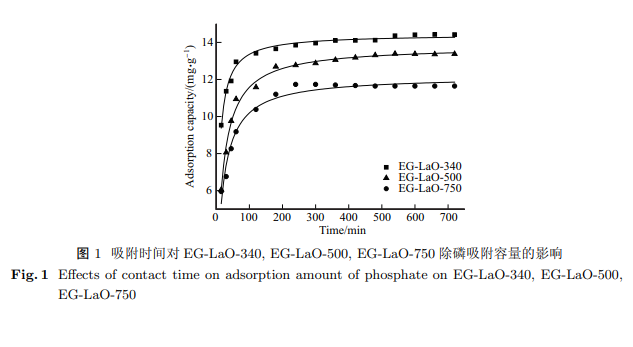
To obtain a highly efficient phosphate removal adsorbent, lanthanum oxide/expanded graphite composite adsorbents were prepared from LaOH load-expanded graphite (EG-LaOH) at 340, 500 and 750 $^\circ$C, namely EG-LaO-340, EG-LaO-500 and EG-LaO-750, respectively. The phosphate removal performances of EG-LaO-340, EG-LaO-500 and EG-LaO-750 were investigated using adsorption kinetics and isotherm experiments. The dephosphorization of the adsorbents was found to be distinctly affected by the preparation temperature, with EG-LaO-340 having the optimal adsorption rate and adsorption capacity. The phosphorus removal mechanism of EG-LaO-340 was investigated by studying its thermodynamics and the pH dependence of adsorption. Additionally, scanning electron microscope (SEM) and Fourier transform infrared (FTIR) spectroscopy were used to characterise the adsorbents. Adsorption was found to be an endothermic and spontaneous process. Electrostatic and Lewis acid-base interactions, in addition to ion exchange, were found to be the main mechanisms of phosphate adsorption.
Key words: phosphorus removal; lanthanum oxide; expanded graphite (EG); adsorption; mechanism
JIN Suwan, NIU Weiya, ZHANG Ling, MAO Xianyong . Preparation, phosphorus removal performance and mechanism of lanthanum oxide/expanded graphite composite adsorbents[J]. Journal of Shanghai University, 2021 , 27(4) : 686 -695 . DOI: 10.12066/j.issn.1007-2861.2176
| [1] | Harke M J, Berry D L, Ammerman J W, et al. Molecular response of the bloom-forming cyanobacterium, microcystis aeruginosa, to phosphorus limitation[J]. Microbial Ecology, 2012, 63(1):188-198. |
| [2] | Choi J W, Kwon K S, Lee S, et al. Pilot-scale test for a phosphate treatment using sulfate-coated zeolite at a sewage disposal facility[J]. Water Air & Soil Pollution, 2014, 225(2):1-13. |
| [3] | Wendling L A, Blomberg P, Sarlin T, et al. Phosphorus sorption and recovery using mineral-based materials: sorption mechanisms and potential phytoavailability[J]. Applied Geochemistry, 2013, 37(10):157-169. |
| [4] | Xiong J, Wang X C, Zhang Q, et al. Characteristics of a landscape water with high salinity in a coastal city of China and measures for eutrophication control[J]. Ecological Indicators, 2015, 61(2):268-273. |
| [5] | Pitakteeratham N, Hafuka A, Satoh H, et al. High efficiency removal of phosphate from water by zirconium sulfate-surfactant micelle mesostructure immobilized on polymer matrix[J]. Water Research, 2013, 47(11):3583-3590. |
| [6] | Su Y, Cui H, Li Q, et al. Strong adsorption of phosphate by amorphous zirconium oxide nanoparticles[J]. Water Research, 2013, 47(14):5018-5026. |
| [7] | Mulkerrins D, Dobson A D W, Colleran E. Parameters affecting biological phosphate removal from wastewaters[J]. Environment International, 2004, 30(2):249-259. |
| [8] | Lu J, Liu D, Jing H, et al. Phosphate removal from aqueous solutions by a nano-structured Fe-Ti bimetal oxide sorbent[J]. Chemical Engineering Research & Design, 2015, 93:652-661. |
| [9] | Wang D, Chen N, Yu Y, et al. Investigation on the adsorption of phosphorus by Fe-loaded ceramic adsorbent[J]. Journal of Colloid & Interface Science, 2015, 464(15):277-284. |
| [10] | Jie X, Yan L, Li C, et al. Removal and recovery of phosphate from water by activated aluminum oxide and lanthanum oxide[J]. Powder Technology, 2015, 269(4):351-357. |
| [11] | Tian S, Jiang P, Ping N, et al. Enhanced adsorption removal of phosphate from water by mixed lanthanum/aluminum pillared montmorillonite[J]. Chemical Engineering Journal, 2009, 151:141-148. |
| [12] | Yoon S Y, Lee C G, Park J A, et al. Kinetic, equilibrium and thermodynamic studies for phosphate adsorption to magnetic iron oxide nanoparticles[J]. Chemical Engineering Journal, 2014, 236:341-347. |
| [13] | Lalley J, Han C, Li X, et al. Phosphate adsorption using modified iron oxide-based sorbents in lake water: kinetics, equilibrium, and column tests[J]. Chemical Engineering Journal, 2016, 284:1386-1396. |
| [14] | Nguyen T A H, Ngo H H, Guo W S, et al. Adsorption of phosphate from aqueous solutions and sewage using zirconium loaded okara (ZLO): fixed-bed column study[J]. Science of the Total Environment, 2015, 523:40-49. |
| [15] | Xie J, Wang Z, Lu S, et al. Removal and recovery of phosphate from water by lanthanum hydroxide materials[J]. Chemical Engineering Journal, 2014, 254:163-170. |
| [16] | Xie J, Zhe W, Da F, et al. Green synjournal of a novel hybrid sorbent of zeolite/lanthanum hydroxide and its application in the removal and recovery of phosphate from water[J]. Journal of Colloid & Interface Science, 2014, 423(3):13-19. |
| [17] | Jiao J H, Wei W, Fenglian S, et al. Highly efficient phosphate scavenger based on well-dispersed La(OH)$_{3}$ nanorods in polyacrylonitrile nanofibers for nutrient-starvation anti-bacterial[J]. ACS Nano, 2015, 9:9292-9302. |
| [18] | 丁文明, 黄霞, 张力平. 水合氧化镧吸附除磷的试验研究[J]. 环境科学, 2003, 24(5):110-113. |
| [18] | Ding W M, Huang X, Zhang L P. Removal of phosphorus from aqueous solution by lanthanum hydrate[J]. Chinese Journal of Environmental Science, 2003, 24(5):110-113. |
| [19] | Ming L C, Chun B H, Yi K L, et al. Selective adsorption and efficient removal of phosphate from aqueous medium with graphene-lanthanum composite[J]. ACS Sustainable Chemistry $\&$ Engineering, 2016, 4(3):1296-1302. |
| [20] | Zhang J, Shen Z, Shan W, et al. Adsorption behavior of phosphate on lanthanum(Ⅲ)-coordinated diamino-functionalized 3D hybrid mesoporous silicates material[J]. Journal of Hazardous Materials, 2011, 186(1):76-83. |
| [21] | Huang Y, Yang J K, Keller A A. Removal of arsenic and phosphate from aqueous solution by metal (hydr-) oxide coated sand[J]. ACS Sustainable Chem Eng, 2014, 2(5):1128-1138. |
| [22] | Huang W. Lanthanum-doped ordered mesoporous hollow silica spheres as novel adsorbents for efficient phosphate removal[J]. Journal of Materials Chemistry A, 2014, 2(23):8839-8848. |
| [23] | Bingcai P, Feichao H, Guangze N, et al. New strategy to enhance phosphate removal from water by hydrous manganese oxide[J]. Environmental Science & Technology, 2014, 48(9):5101-5107. |
| [24] | Chen N, Feng C, Zhang Z, et al. Preparation and characterization of lanthanum(Ⅲ) loaded granular ceramic for phosphorus adsorption from aqueous solution[J]. Journal of the Taiwan Institute of Chemical Engineers, 2012, 43(5):783-789. |
| [25] | Irene E, Jie Y, Jun Z, et al. Low-cost and large-scale synjournal of functional porous materials for phosphate removal with high performance[J]. Nanoscale, 2013, 5(13):6173-6180. |
| [26] | Feng S Z, Qi Z, Xu Y, et al. Rapid preparation of expanded graphite by microwave irradiation for the extraction of triazine herbicides in milk samples[J]. Food Chemistry, 2016, 197:943-949. |
| [27] | Zhang L, Gao Y, Li M, et al. Expanded graphite loaded with lanthanum oxide used as a novel adsorbent for phosphate removal from water: performance and mechanism study[J]. Environmental Technology, 2015, 36(8):1016-1025. |
| [28] | 苏阳, 靳苏皖, 张玲. 负载氢氧化镧的膨胀石墨除磷剂的制备、性能及再生[J]. 上海大学学报(自然科学版), 2016, 22(6):776-783. |
| [28] | Su Y, Jin S W, Zhang L. Preparation, performance and recovery of expanded graphite loaded with lanthanum hydroxide[J]. Journal of Shanghai University (Natural Science Edition), 2016, 22(6):776-783. |
| [29] | Ozawa M, Onoe R, Kato H. Formation and decomposition of some rare earth (RE=La, Ce, Pr) hydroxides and oxides by homogeneous precipitation[J]. Journal of Alloys & Compounds, 2006, 408(2):556-559. |
| [30] | Shi Z L, Liu F M, Yao S H. Adsorptive removal of phosphate from aqueous solutions using activated carbon loaded with Fe(Ⅲ) oxide[J]. New Carbon Materials, 2011, 26(4):299-306. |
| [31] | Biswas B K, Inoue K, Ghimire K N, et al. The adsorption of phosphate from an aquatic environment using metal-loaded orange waste[J]. Journal of Colloid & Interface Science, 2007, 312(2):214-223. |
| [32] | Yan L G, Xu Y Y, Yu H Q, et al. Adsorption of phosphate from aqueous solution by hydroxy-aluminum, hydroxy-iron and hydroxy-iron-aluminum pillared bentonites[J]. Journal of Hazardous Materials, 2010, 179:244-250. |
| [33] | Yang J, Zhou L, Zhao L, et al. A designed nanoporous material for phosphate removal with high efficiency[J]. Journal of Materials Chemistry A, 2011, 21(8):2489-2494. |
| [34] | Wang Z, Shen D, Fei S, et al. Phosphate adsorption on lanthanum loaded biochar[J]. Chemosphere, 2016, 150:1-7. |
| [35] | Aghazadeh M, Golikand A N, Ghaemi M, et al. A novel lanthanum hydroxide nanostructure prepared by cathodic electrodeposition[J]. Materials Letters, 2011, 65(10):1466-1468. |
| [36] | Wang Z, Fan Y, Li Y, et al. Synjournal of zeolite/hydrous lanthanum oxide composite from coal fly ash for efficient phosphate removal from lake water[J]. Microporous & Mesoporous Materials, 2015, 222:226-234. |
| [37] | Rivera-Utrilla J, Sánchez-Polo M, Bautista-Toledo M I, et al. Enhanced oxidation of sodium dodecylbenzenesulfonate aqueous solution using ozonation catalyzed by base treated zeolite[J]. Chemical Engineering Journal, 2012, 180(6):204-209. |
| [38] | Zhang L, Zhou Q, Liu J, et al. Phosphate adsorption on lanthanum hydroxide-doped activated carbon fiber[J]. Chemical Engineering Journal, 2012, 185(6):160-167. |
| [39] | Zhang L, Gao Y, Zhou Q, et al. High-performance removal of phosphate from water by graphene nanosheets supported lanthanum hydroxide nanoparticles[J]. Water Air & Soil Pollution, 2014, 225(6):1-11. |
| [40] | Fei L, Gong J L, Zeng G M, et al. Removal of phosphate from aqueous solution by magnetic Fe-Zr binary oxide[J]. Chemical Engineering Journal, 2011, 171(2):448-455. |
/
| 〈 |
|
〉 |