

Journal of Shanghai University >
Syntheses and Photochromic properties of 4-fluoroacetophenone disubstituted viologen
Accepted date: 2021-05-07
Online published: 2021-05-28
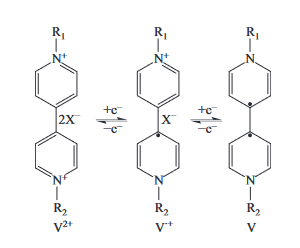
Key words: viologen derivative; photochromism; viologen radical
ZHANG Haotian, BAI YueLing, HANG Xiangcao, AN BaoLi, XU Jiaqiang . Syntheses and Photochromic properties of 4-fluoroacetophenone disubstituted viologen[J]. Journal of Shanghai University, 2024 , 30(1) : 68 -076 . DOI: 10.12066/j.issn.1007-2861.2301
/
| 〈 |
|
〉 |