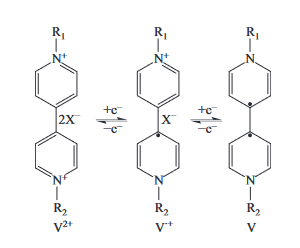
紫精材料在外界刺激 (如光、热、电) 下会表现出灵敏的变色行为, 在光学开关和分子传感识别中具有良好的潜在应用前景. 通过一步反应合成了 4-氟苯乙酮双取代紫精衍生
物 (VioF2·2Cl). 氟苯乙酮取代基的引入扩大了其共轭体系, 起到稳定自由基的作用, 使着色
更稳定持久. VioF2·2Cl 在紫外 (ultraviolet, UV) 灯的照射下可以快速地从白色转变为浅黄
色直至黄绿色, 显示出了灵敏的光致变色行为. 变色后晶体中的 VioF22+ 分子间具有更短的
π · · · π∗ 作用距离, 能够促进光致电子转移 (photoinduced electron transfer, PET) 过程的发
生, 并且起到稳定紫精阳离子自由基的作用, 使其能够在氧气环境下持续变色 3 d 而不被氧化
褪色. 这种新型的紫精衍生物表现出了更为灵敏的光致变色性能及自由基着色稳定性, 在光致
变色材料领域具有良好的潜在应用可能.
Owing to their sensitive chromic behaviour under external stimulation, such
as light, heat, and electricity, viologen-based materials have shown considerable potential
in optical switching and molecular sensor recognition applications. 4-Fluoroacetophenone
disubstituted viologen (VioF2·2Cl) was synthesised via a one-step reaction. The introduction of fluoroacetophenone substituents expands the conjugated system of the molecule
which stabilises the radical structure and facilitates retention of the colour change for a
longer period of time. The synthesized VioF2·2Cl exhibited sensitive photochromic properties, showing a rapid colour change from white to light yellow and then to yellow-green
under ultraviolet (UV) light irradiation. Analysis of the crystal structure confirmed that
the VioF2+
2 molecules possess a shorter π · · · π∗ interaction distance after discoloration,
which induces photoinduced electron transfer (PET) and thus stabilizes the viologen radical. VioF2·2Cl crystals can maintain their coloured radical state in an oxygen environment
for three days without significant fading. This novel viologen derivative exhibits better sen-
sitive photochromic properties and free radical colour stability essential for its application in the field of photochromic materials.