

收稿日期: 2019-03-27
网络出版日期: 2019-05-28
基金资助
国家自然科学基金资助项目(30971095)
MEK5$\alpha $ and MEK5$\beta $ differentially regulate Beclin 1 promoter
Received date: 2019-03-27
Online published: 2019-05-28
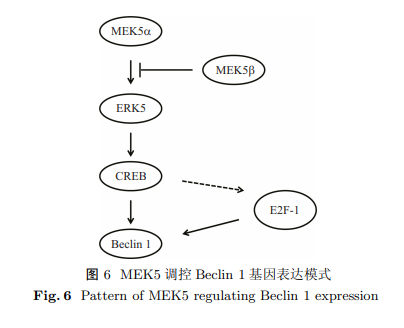
Beclin 1是哺乳动物自噬相关基因, 调控自噬起始和自噬体成熟. 在肌肉分化过程中, Beclin 1 基因表达上调, 自噬增加; 此外 MEK5-ERK5 信号活化并调控成肌细胞分化. 因此, 在肌肉分化过程中, MEK5-ERK5 信号通路可能调控 Beclin 1 基因表达. 目的是阐明 MEK5 对成肌细胞 Beclin 1 基因启动子活性的调控. 将不同长度 Beclin 1 启动子片段克隆至荧光素酶报告基因载体pGL3-Basic并转染成肌细胞C2C12. 双荧光素酶报告基因检测实验结果显示, 含 Beclin 1 基因起始密码子上游 586 碱基对 DNA 片段的载体(p-354)具有强荧光素酶活性. MEK5$\alpha $显著增加 p-354 荧光素酶活性, 并有剂量依赖性; 而 MEK5$\beta $ 显著降低 p-354 荧光素酶活性. MEK5$\beta $能够拮抗 MEK5$\alpha $对 p-354 荧光素酶活性的调控. 与 MEK5 对 Beclin 1 基因启动子调控结果一致, MEK5$\alpha $CA 上调细胞Beclin 1 mRNA 表达, MEK5$\beta $DD 下调 Beclin 1 mRNA 表达, 并且 MEK5$\beta $DD 抑制 MEK5$\alpha $CA 对 Beclin 1 mRNA 表达的促进作用. 此外, 转录因子 CREB 家族成员 CREB3, CREBP 和 CREBL1 能够显著上调 p-354 荧光素酶活性. CREB3 呈剂量依赖性显著上调 p-354 荧光素酶活性, 并与 MEK5$\alpha $ 具有协同效应. MEK5$\alpha $ 和 MEK5$\beta $ 对 Beclin 1 启动子具有不同调控作用, CREB 可能是其下游效应因子.
刘晓芸, 王雷斌, 王庆, 张沙沙, 赵微苗, 贺林, 朱洪新 . MEK5$\alpha $ 和 MEK5$\beta $ 调控 ${\bf Beclin}$ 1 启动子[J]. 上海大学学报(自然科学版), 2021 , 27(3) : 563 -572 . DOI: 10.12066/j.issn.1007-2861.2142
Beclin 1, a mammalian autophagy-related gene, regulates autophagy initiation and autophagosome maturation. During muscle differentiation, Beclin 1 is upregulated. Additionally, MEK5-ERK5 is activated to regulate muscle differentiation. Thus, MEK5-ERK5 may regulate Beclin 1 gene expression during muscle differentiation. The aim of this study was to determine MEK5 regulation of Beclin 1 promoter in myoblast cells. A series of promoter-luciferase constructs harbouring different lengths of Beclin 1 promoter were created and transfected into myoblast C2C12 cells. In the luciferase assay, the construct containing 586 base pairs upstream of the start codon (p-354) exhibited the most potent luciferase activity. MEK5$\alpha $ and MEK5$\beta $ enhanced and suppressed p-354 luciferase activity, respectively. MEK5$\beta $ is capable of antagonizing the effect of MEK5$\alpha $ on p-354 luciferase activity. Consistent with the results on the regulatory effects of MEK5 on Beclin 1 promoter, MEK5$\alpha $CA and MEK5$\beta $DD upregulated and downregulated Beclin 1 mRNA expression, respectively. Moreover, MEK5$\beta $DD antagonised the stimulatory effects of MEK5$\alpha $CA on Beclin 1 mRNA expression. Members of the CREB family, including CREB3, CREBP, and CREBL1, promoted p-354 luciferase activity. Furthermore, CREB3 dose-dependently increased p-354 luciferase activity and exhibited a synergistic effect with MEK5$\alpha $ on p-354 luciferase activity. Collectively, these findings indicate that MEK5$\alpha $ and MEK5$\beta $ differentially regulate Beclin 1 promoter activity and that CREB family members may be downstream effectors.

Key words: MEK5; Beclin 1; promoter; luciferase reporter gene
| [1] | Klionsky D J, Emr S D. Autophagy as a regulated pathway of cellular degradation[J]. Science, 2000,290(17):17-21. |
| [2] | Dikic I, Elazar Z. Mechanism and medical implications of mammalian autophagy[J]. Nat Rev Mol Cell Biol, 2018,19:349-364. |
| [3] | Liang X H, Jackson S, Seaman M, et al. Induction of autophagy and inhibition of tumorigenesis by beclin 1[J]. Nature, 1999,402:672-676. |
| [4] | Aita V M, Liang X H, Murty V V, et al. Cloning and genomic organization of Beclin 1, a candidate tumor suppressor gene on chromosome 17q21[J]. Genomics, 1999,59:59-65. |
| [5] | Hill S M, Wrobel L, Rubinsztein D C. Post-translational modifications of Beclin 1 provide multiple strategies for autophagy regulation[J]. Cell Death Differ, 2019,26:617-629. |
| [6] | English J M, Vanderbilt C A, Xu S, et al. Isolation of MEK5 and differential expression of alternatively spliced forms[J]. J Biol Chem, 1995,270:28897-28902. |
| [7] | Cameron S J, Abe J, Malik S, et al. Differential role of MEK5alpha and MEK5beta in BMK1/ERK5 activation[J]. J Biol Chem, 2004,279:1506-1512. |
| [8] | Gu J, Hu W, Song Z P, et al. Rapamycin inhibits cardiac hypertrophy by promoting autophagy via the MEK/ERK/Beclin-1 pathway[J]. Front Physiol, 2016, DOI: 10.3389/fphys.2016.00104. |
| [9] | Wang J, Whiteman M W, Lian H, et al. A non-canonical MEK/ERK signaling pathway regulates autophagy via regulating Beclin 1[J]. J Biol Chem, 2009,284:21412-21424. |
| [10] | Li D D, Wang L L, Deng R, et al. The pivotal role of c-Jun NH2-terminal kinase-mediated Beclin 1 expression during anticancer agents-induced autophagy in cancer cells[J]. Oncogene, 2009,28:886-898. |
| [11] | Park S, Choi Y, Jung N, et al. Autophagy induction in the skeletal myogenic differentiation of human tonsil-derived mesenchymal stem cells[J]. Int J Mol Med, 2017,39:831-840. |
| [12] | Gamberi T, Modesti A, Magherini F, et al. Activation of autophagy by globular adiponectin is required for muscle differentiation[J]. Biochim Biophys Acta, 2016,1863:694-702. |
| [13] | Fortini P, Ferretti C, Iorio E, et al. The fine tuning of metabolism, autophagy and differentiation during in vitro myogenesis[J]. Cell Death Dis, 2016,7(3):e2168. |
| [14] | Carter E J, Cosgrove R A, Gonzalez I, et al. MEK5 and ERK5 are mediators of the pro-myogenic actions of IGF-2[J]. J Cell Sci, 2009,122:3104-3112. |
| [15] | Chen T H, Chen C Y, Wen H C, et al. YAP promotes myogenic differentiation via the MEK5-ERK5 pathway[J]. FASEB J, 2017,31:2963-2972. |
| [16] | Dinev D, Jordan B W, Neufeld B, et al. Extracellular signal regulated kinase 5 (ERK5) is required for the differentiation of muscle cells[J]. EMBO Rep, 2001,2:829-834. |
| [17] | Watson F L, Heerssen H M, Bhattacharyya A, et al. Neurotrophins use the Erk5 pathway to mediate a retrograde survival response[J]. Nat Neurosci, 2001,4:981-988. |
| [18] | Weinmann A S, Bartley S M, Zhang T, et al. Use of chromatin immunoprecipitation to clone novel E2F target promoters[J]. Mol Cell Biol, 2001,21:6820-6832. |
| [19] | Melnikova V O, Dobroff A S, Zigler M, et al. CREB inhibits AP-2alpha expression to regulate the malignant phenotype of melanoma[J]. PLoS One, 2010,5:e12452. |
| [20] | Wan B, Zang Y, Wang L. Overexpression of Beclin 1 inhibits proliferation and promotes apoptosis of human laryngeal squamous carcinoma cell Hep-2[J]. Onco Targets Ther, 2018,11:3827-3833. |
| [21] | Wechman S L, Pradhan A K, DeSalle R, et al. New insights into Beclin-1: evolution and pan-malignancy inhibitor activity[J]. Adv Cancer Res, 2018,137:77-114. |
| [22] | Zhu H, He L. Beclin 1 biology and its role in heart disease[J]. Curr Cardiol Rev, 2015,11:229-237. |
| [23] | Ejlerskov P, Ashkenazi A, Rubinsztein D C. Genetic enhancement of macroautophagy in vertebrate models of neurodegenerative diseases[J]. Neurobiol Dis, 2019,122:3-8. |
/
| 〈 |
|
〉 |