

收稿日期: 2019-03-18
网络出版日期: 2019-02-04
基金资助
低碳转化科学与工程重点实验室开放课题资助项目(KLLCCSE-201707);低碳转化科学与工程重点实验室开放课题资助项目(SARI);低碳转化科学与工程重点实验室开放课题资助项目(CAS)
Preparation of lactic acid by catalytic conversion of glucose by dual-metal ions
Received date: 2019-03-18
Online published: 2019-02-04
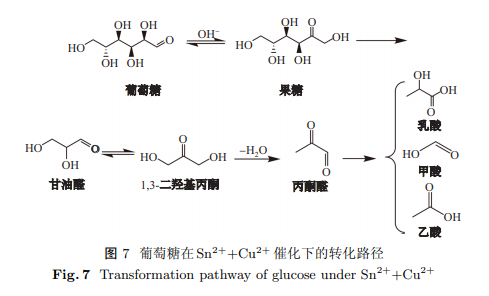
选择不同的双金属阳离子作为催化剂, 在碱性条件下用于水热转化葡萄糖制备乳酸, 发现Sn$^{2+}$和Cu$^{2+}$具有一定的协同作用. 研究了反应温度、时间、碱加入量和金属离子比例对乳酸产率的影响, 获得最优工艺条件如下: 0.025 g葡萄糖, 2 mmolNaOH, $n$(Sn$^{2+})$:$n$(Cu$^{2+})$=0.25:0.75(共1 mmol), 20 mL水, 200 ${^\circ}$C反应温度, 3 h反应时间. 在最优条件下, 乳酸产率为64.1%, 甲酸产率为20.3%, 乙酸产率为11%. 选用几种模型化合物作为反应的中间产物进行水热转化实验, 并根据实验结果对水热转化葡萄糖的转化机理进行了探究. 结果发现, 转化过程中重要的中间产物为果糖、1,3-二羟基丙酮和丙酮醛. 两种金属离子存在协同作用, Cu$^{2+}$抑制了乳酸的分解, Sn$^{2+}$提高了丙酮醛转化为乳酸的转化效率, 最终提高了乳酸产率.
李航伟, 杨翔, 罗小飞, 郭晓亚, 孔令照 . 双金属离子水热催化转化葡萄糖制备乳酸[J]. 上海大学学报(自然科学版), 2021 , 27(2) : 379 -388 . DOI: 10.12066/j.issn.1007-2861.2152
Different dual-metal ions were selected as catalysts to prepare lactic acid (LA) by hydrothermal conversion of glucose under alkaline conditions, and Sn$^{2+}$ and Cu$^{2+}$ were found to have a certain synergistic effect. The effects of reaction temperature, time, alkali addition amount, and metal ion ratio on lactic acid yield were studied to optimise the experimental conditions. The optimum process conditions were as follows: 0.025 g glucose, 2 mmol sodium hydroxide, $n$(Sn$^{2+})$:$n$(Cu$^{2+})$=0.25:0.75 (1 mmol total), 20 mL water, reaction temperature 200 ${^\circ}$C, and reaction time 3 h. Under optimal conditions, the yields of lactic acid, formic acid (FA), and acetic acid (AA) were 64.1%, 20.3% and 11%, respectively. Several model compounds were selected as intermediate products for the hydrothermal transformation experiments. According to the experimental results, the conversion mechanism of the hydrothermal conversion of glucose was investigated. Important intermediates detected in the conversion process were fructose, 1,3-dihydroxyacetone and pyruvaldehyde. There was a synergistic effect between the two metal ions, as Cu$^{2+}$ inhibited the decomposition of lactic acid, and Sn$^{2+}$ increased the conversion efficiency of pyruvaldehyde to lactic acid, finally increasing the yield of lactic acid.

Key words: metal ion; reaction mechanism; lactic acid (LA); 1,3-dihydroxyacetone; pyruvaldehyde
| [1] | Rathin D, Michael H. Lactic acid: recent advances in products, processes and technologies: a review[J]. J Chem Technol Biotechnol, 2006,81(7):1119-1129. |
| [2] | Fan Y X, Zhou C H, Zhu X H. Selective catalysis of lactic acid to produce commodity chemicals[J]. Catalysis Reviews, 2009,51(3):293-324. |
| [3] | Zhang Y X, Yoshida M, Vadlani P V. Biosynjournal of D-lactic acid from lignocellulosic biomass[J]. Biotechnology Letters, 2018,40(8):1167-1179. |
| [4] | Marianou A A, Michailofa C M, Pineda A, et al. Effect of Lewis and Br?nsted acidity on glucose conversion to 5-HMF and lactic acid in aqueous and organic media[J]. Applied Catalysis A(General), 2018,555:75-87. |
| [5] | Verziu M, Serano M, Jurca B, et al. Catalytic features of Nb-based nanoscopic inorganic fluorides for an efficient one-pot conversion of cellulose to lactic acid[J]. Catalysis Today, 2018,306:102-110. |
| [6] | Wang S W, Chen K Y, Wang Q B. Ytterbium triflate immobilized on sulfo-functionalized SBA-15 catalyzed conversion of cellulose to lactic acid[J]. Journal of Porous Materials, 2018,25(5):1531-1539. |
| [7] | Onda A, Ochi T, Kajiyoshi K, et al. A new chemical process for catalytic conversion of D-glucose into lactic acid and gluconic acid[J]. Applied Catalysis A(General), 2008,343(1/2):49-54. |
| [8] | Zhang S P, Jin F M, Hu J J, et al. Role of metallic Zn, Ni and activated carbon additives in improving the hydrothermal conversion of glucose into lactic acid[J]. J Chem Technol Biotechnol, 2017,92(5):1046-1052. |
| [9] | Wattanapaphawong P, Sato O, Sato K, et al. Conversion of cellulose to lactic acid by using ZrO$_{2}$-Al$_{2}$O$_{3}$ catalysts[J]. Catalysts, 2017,7(221):2-10. |
| [10] | Onda A, Ochi T, Kajiyoshi K, et al. Lactic acid production from glucose over activated hydrotalcites as solid base catalysts in water[J]. Catalysis Communications, 2008,9(6):1050-1053. |
| [11] | Xue Y, Jin F M, Yoshikaw K. Hydrothermal lactic acid production from glucose over feldspars as solid base catalysts in water[J]. Energy Procedia, 2014,61:2474-2477. |
| [12] | Cao D, Cai W J, Tao W T, et al. Lactic acid production from glucose over a novel Nb$_{2}$O$_{5}$ nanorod catalyst[J]. Catal Lett, 2017,147(4):926-933. |
| [13] | Holm M S, Saravanamurugan S, Taarning E. Conversion of sugars to lactic acid derivatives using heterogeneous zeotype catalysts[J]. Science, 2010,328(5978):603-605. |
| [14] | Lau K S, Chia C H, Chin S X, et al. Conversion of glucose into lactic acid using silica-supported zinc oxide as solid acid catalyst[J]. Pure Appl Chem, 2018,90(6):1035-1043. |
| [15] | Wattanapaphawong P, Reubroycharonebc P, Yamaguch A. Conversion of cellulose into lactic acid using zirconium oxide catalysts[J]. RSC Adv, 2017,7(30):18561-18568. |
| [16] | Huo Z B, Fang Y, Ren D Z, et al. Selective conversion of glucose into lactic acid with transition metal ions in diluted aqueous NaOH solution[J]. ACS Sustainable Chem Eng, 2014,2(12):2765-2771. |
| [17] | Wang Y L, Deng W P, Wang B J, et al. Chemical synjournal of lactic acid from cellulose catalysed by lead(Ⅱ) ions in water[J]. Nature Communications, 2013,4(1):1-7. |
| [18] | Yan X Y, Jin F M, Tohji K, et al. Production of lactic acid from glucose by alkaline hydrothermal reaction[J]. J Mater Sci, 2007,42(24):9995-9999. |
| [19] | Li L Y, Shen F, Smithc R L, et al. Quantitative chemocatalytic production of lactic acid from glucose under anaerobic conditions at room temperature[J]. Green Chem, 2017,19(1):76-81. |
| [20] | Bayu A, Yoshida A, Karnjanakom S, et al. Catalytic conversion of biomass derivatives to lactic acid with increased selectivity in an aqueous tin(Ⅱ) chloride/choline chloride system[J]. Green Chem, 2018,20:4112-4119. |
| [21] | Hayashi Y, Sasaki Y. Tin-catalyzed conversion of trioses to alkyl lactates in alcohol solution[J]. Chem Commun, 2005,36(41):2716-2718. |
| [22] | Rasrendra C B, Fachri B A, Makertihartha I, et al. Catalytic conversion of dihydroxyacetone to lactic acid using metal salts in water[J]. ChemSusChem, 2011,4(6):768-777. |
| [23] | Wang X C, Song Y L, Huang C P, et al. Lactic acid production from glucose over polymer catalysts in aqueous alkaline solution under mild conditions[J]. Green Chem, 2014,16(9):4234-4240. |
| [24] | Wang Y Q, Jin F M, Sasaki M, et al. Selective conversion of glucose into lactic acid and acetic acid with copper oxide under hydrothermal conditions[J]. AIChE Journal, 2013,59(6):2096-2104. |
| [25] | Lawrence G D, Mavi A, Meral K. Promotion by phosphate of Fe(Ⅲ)- and Cu(Ⅱ)-catalyzed autoxidation of fructose[J]. Carbohydr Res, 2008,343(4):626-635. |
/
| 〈 |
|
〉 |