

MnO@CoMn$_{\textbf{2}}$O$_{\textbf{4}}$/N-C纳米线复合材料的制备及其作为锂离子电池阳极材料的电化学性能
收稿日期: 2019-03-18
网络出版日期: 2019-10-28
基金资助
国家自然科学基金资助项目(21601120);安徽省高校自然科学基金资助项目(KJ2016A510);安徽省高校优秀青年人才基金资助项目(gxyq2017104)
Preparation and electrochemical properties as anode materials for lithium-ion batteries of MnO@CoMn$_{\textbf{2}}$O$_{\textbf{4}}$/N-C nanowire composites
Received date: 2019-03-18
Online published: 2019-10-28
过渡金属氧化物作为锂离子电池(lithium-ion batteries, LIBs)阳极材料时具有较高的理论容量, 但因其电导率低, 以及充放电过程中的体积膨胀效应常会导致容量的快速衰减. 碳包覆是提升金属氧化物导电性的有效方法, 二者之间的协同效应也可以有效提升材料的电化学性能. 以MnO$_{2}$纳米线为模板制备出MnO$_{2}$@ZIF-67有机-无机杂化纳米结构, 再通过退火处理合成了氮掺杂碳包覆的MnO@CoMn$_{2}$O$_{4}$纳米线复合材料(MnO@CoMn$_{2}$O$_{4}$@N-C). ZIF-67的有机配体在高温煅烧过程中发生碳化反应, 产生了氮掺杂碳, 提升了导电性. 当作为锂离子电池阳极材料时, MnO@CoMn$_{2}$O$_{4}$/N-C纳米线复合材料在0.1 A/g电流密度下的首次放电比容量为1 594.6 mA$\cdot$h/g, 并且在100次充放电循环后的放电比容量仍保持在 925.8 mA$\cdot$h/g, 在0.5 A/g电流密度下经200次充放电循环后的放电比容量仍维持在837.6 mA$\cdot$h/g, 同时具有优异的倍率循环性能. 这种优异的电化学储能特性主要来源于复合材料的特殊结构, 以及氮掺杂碳的包覆.
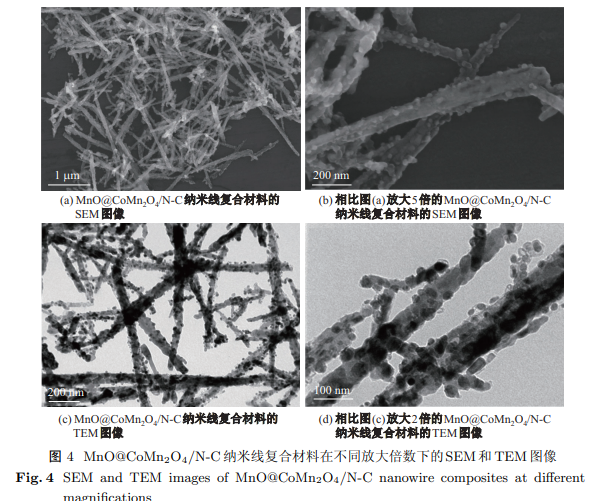
程二波, 王尚岱, 黄守双, 陈大勇, 胡张军, 陈志文 . MnO@CoMn$_{\textbf{2}}$O$_{\textbf{4}}$/N-C纳米线复合材料的制备及其作为锂离子电池阳极材料的电化学性能[J]. 上海大学学报(自然科学版), 2021 , 27(2) : 400 -410 . DOI: 10.12066/j.issn.1007-2861.2155
Transition metal oxides have a higher theoretical capacity as anode materials for lithium-ion batteries (LIBs). However, they often suffer from poor capacity retention owing to their low electrical conductivity and large volume variations during the charge/discharge process. Carbon coating is an effective way to enhance the electrical conductivity of metal oxides. The synergistic effects of the two components can also effectively improve the electrochemical performance of the material. In this study, MnO$_{2}$@ZIF-67 organic-inorganic hybrid nanostructures were prepared using MnO$_{2}$ nanowires as a template. N-doped carbon-coated MnO@CoMn$_{2}$O$_{4}$ nanowire composites (MnO@CoMn$_{2}$O$_{4}$@N-C) were synthesised by annealing. The organic ligand of ZIF-67 underwent carbonisation during high-temperature calcination to produce N-doped carbon, which improved the electrical conductivity. When MnO@CoMn$_{2}$O$_{4}$/N-C nanowire composites were used as the anode material for LIBs, a discharge capacity of 1 594.6 mA$\cdot$h/g was achieved in the first cycle, with a retention capacity of 925.8 mA$\cdot$h/g even after 100 cycles at current density of 0.1 A/g and a reversible capacity of 837.6 mA$\cdot$h/g at 0.5 A/g. It also had an excellent rate cycling performance. The outstanding electrochemical performance was attributed to the unique structure as well as the coating effect of N-doped carbon.

| [1] | Yang Z G, Zhang J L, Kintner-Meyer M C W, et al. Electrochemical energy storage for green grid[J]. Chemical Review, 2011,111(5):3577-3613. |
| [2] | Zhong Y R, Yang M, Zhon X L, et al. Structural design for anodes of lithium-ion batteries: emerging horizons from materials to electrodes[J]. Materials Horizons, 2015,2(6):553-566. |
| [3] | Lu J, Chen Z W, Pan F, et al. High-performance anode materials for rechargeable lithium-ion batteries[J]. Electrochemical Energy Reviews, 2018,1(1):35-53. |
| [4] | Guo Y, Yu L, Wang C Y, et al. Hierarchical tubular structures composed of Mn-based mixed metal oxide nanoflakes with enhanced electrochemical properties[J]. Advance Function Materials, 2015,25(32):5184-5189. |
| [5] | Ge X L, Li Z Q, Wang C X, et al. Metal-organic frameworks derived porous core/shell structured ZnO/ZnCo$_{2}$O$_{4}$/C hybrids as anodes for high-performance lithium-ion battery[J]. ACS Appliled Materials Interfaces, 2015,7(48):26633-26642. |
| [6] | Cai Z, Xu L, Yan M, et al. Manganese oxide/carbon yolk-shell nanorod anodes for high capacity lithium batteries[J]. Nano Letters, 2015,15(1):738-744. |
| [7] | Chen J, Wu X, Gong Y, et al. General synjournal of transition-metal oxide hollow nanospheres/nitrogen-doped graphene hybrids by metal-ammine complex chemistry for high-performance lithium-ion batteries[J]. Chemistry, 2018,24(9):2126-2136. |
| [8] | Lu Y, Yu Y, Lou X W. Nanostructured conversion-type anode materials for advanced lithium-ion batteries[J]. Chem-Us, 2018,4(5):972-996. |
| [9] | Xiao Y, Cao M H. Carbon-anchored MnO nanosheets as an anode for high-rate and long-life lithium-ion batteries[J]. ACS Applied Materials Interfaces, 2015,7(23):12840-12849. |
| [10] | Xia Y, Xiao Z, Dou X, et al. Green and facile fabrication of hollow porous MnO/C microspheres from microalgaes for lithium-ion batteries[J]. ACS Nano, 2013,7(8):7083-7092. |
| [11] | Zhong M, Yang D, Xie C, et al. Yolk-shell MnO@ZnMn$_{2}$O$_{4}$/N-C nanorods derived from alpha-MnO$_{2}$/ZIF-8 as anode materials for lithium ion batteries[J]. Small, 2016,12(40):5564-5571. |
| [12] | Zhang H, Liu X M, Wu Y, et al. MOF-derived nanohybrids for electrocatalysis and energy storage: current status and perspectives[J]. Chemical Communications, 2018,54(42):5268-5288. |
| [13] | Salunkhe R R, Kaneti Y V, Kim J, et al. Nanoarchitectures for metal-organic framework-derived nanoporous carbons toward supercapacitor applications[J]. Accounts of Chemical Research, 2016,49(12):2796-2806. |
| [14] | Garcia-Garcia P, Muller M, Corma A. MOF catalysis in relation to their homogeneous counterparts and conventional solid catalysts[J]. Chemical Science, 2014,5(8):2979-3007. |
| [15] | Zhang H, Wang Y, Zhao W, et al. MOF-derived ZnO nanoparticles covered by N-doped carbon layers and hybridized on carbon nanotubes for lithium-ion battery anodes[J]. ACS Applied Materials Interfaces, 2017,9(43):37813-37822. |
| [16] | Rowsell J L C, Yaghi O M. Metal-organic frameworks: a new class of porous materials[J]. Microporous and Mesoporous Materials, 2004,73(1/2):3-14. |
| [17] | Kreno L E, Leong K, Farha O K, et al. Metal-organic framework materials as chemical sensors[J]. Chemical Review, 2012,112(2):1105-1125. |
| [18] | Furukawa H, Cordova K E, O'keeffe M, et al. The chemistry and applications of metal-organic frameworks[J]. Science, 2013,341(6149):1230444. |
| [19] | Murray L J, Dinca M, Long J R. Hydrogen storage in metal-organic frameworks[J]. Chemical Society Review, 2009,38(5):1294-1314. |
| [20] | Jiang H L, Liu B, Lan Y Q, et al. From metal-organic framework to nanoporous carbon: toward a very high surface area and hydrogen uptake[J]. Journal of American Chemical Society, 2011,133(31):11854-11857. |
| [21] | Li Z X, Zou K Y, Zhang X, et al. Hierarchically flower-like N-doped porous carbon materials derived from an explosive 3-fold interpenetrating diamondoid copper metal-organic framework for a supercapacitor[J]. Inorganic Chemistry, 2016,55(13):6552-6562. |
| [22] | Zheng F C, Yang Y, Chen Q W. High lithium anodic performance of highly nitrogen-doped porous carbon prepared from a metal-organic framework[J]. Nature Communications, 2014,5:5261. |
| [23] | Zhang L M, Yan B, Zhang J H, et al. Design and self-assembly of metal-organic framework-derived porous Co$_{3}$O$_{4}$ hierarchical structures for lithium-ion batteries[J]. Ceramics International 2016,42(4):5160-5170. |
| [24] | Zou F, Hu X, Li Z, et al. MOF-derived porous ZnO/ZnFe$_{2}$O$_{4}$/C octahedra with hollow interiors for high-rate lithium-ion batteries[J]. Advance Materials, 2014,26(38):6622-6628. |
| [25] | Yang S J, Kim T, Im J H, et al. MOF-derived hierarchically porous carbon with exceptional porosity and hydrogen storage capacity[J]. Chemical Materials, 2012,24(3):464-470. |
| [26] | 林佳, 林晓明, 石光, 等. MOFs 作为模板制备锂离子电池负极材料的研究进展[J]. 科学通报, 2018(16):1538-1549. |
| [26] | Lin J, Lin X M, Shi G, et al. Research progress of MOFs as template for the preparation of anode materials for lithium ion batteries[J]. Chinese Science Bulletin, 2018(16):1538-1549. |
| [27] | Huang M, Mi K, Zhang J H, et al. MOF-derived bi-metal embedded N-doped carbon polyhedral nanocages with enhanced lithium storage[J]. Journal of Materials Chemistry A, 2017,5(1):266-274. |
/
| 〈 |
|
〉 |